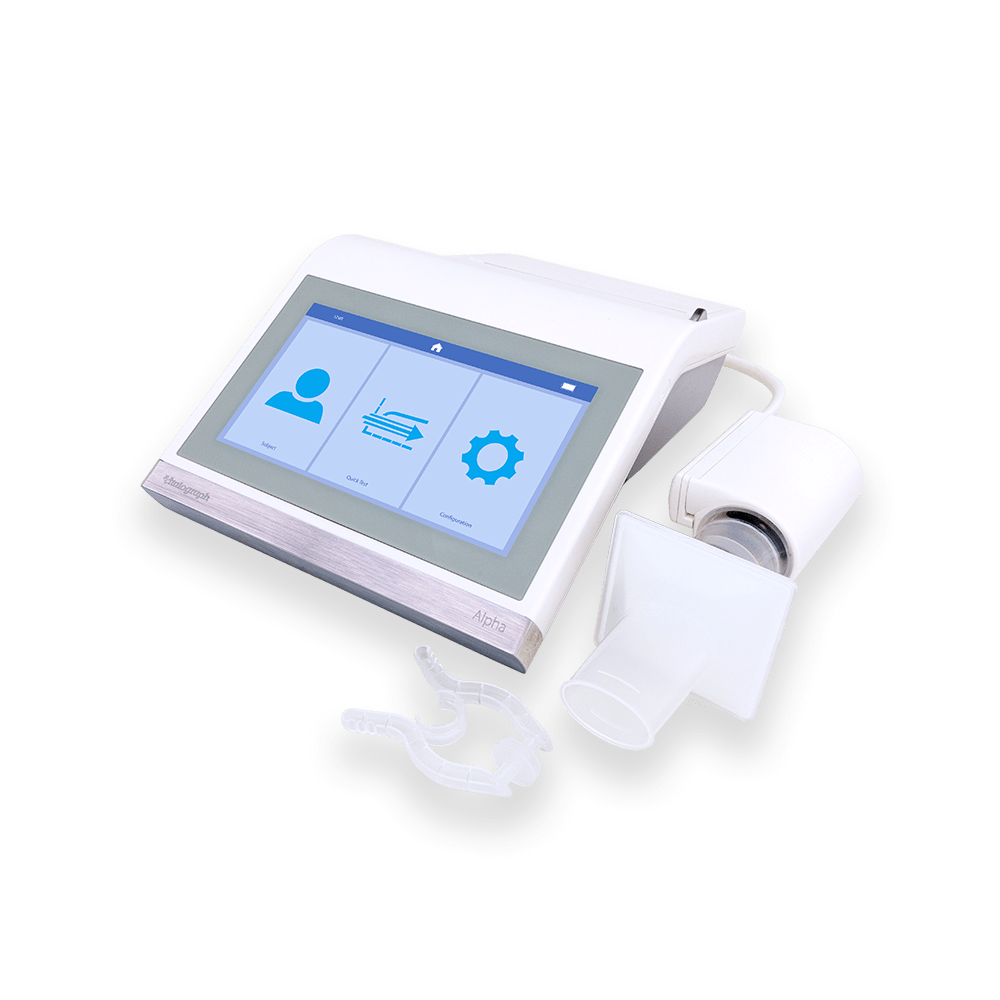
Alpha Spirometer
Highly portable lightweight all-in-one desktop spirometer and printer. Easy to use and no PC required.
$1.00
Designed for testing in a clinic or on the go, the Alpha is our all-in-one desktop spirometry solution.
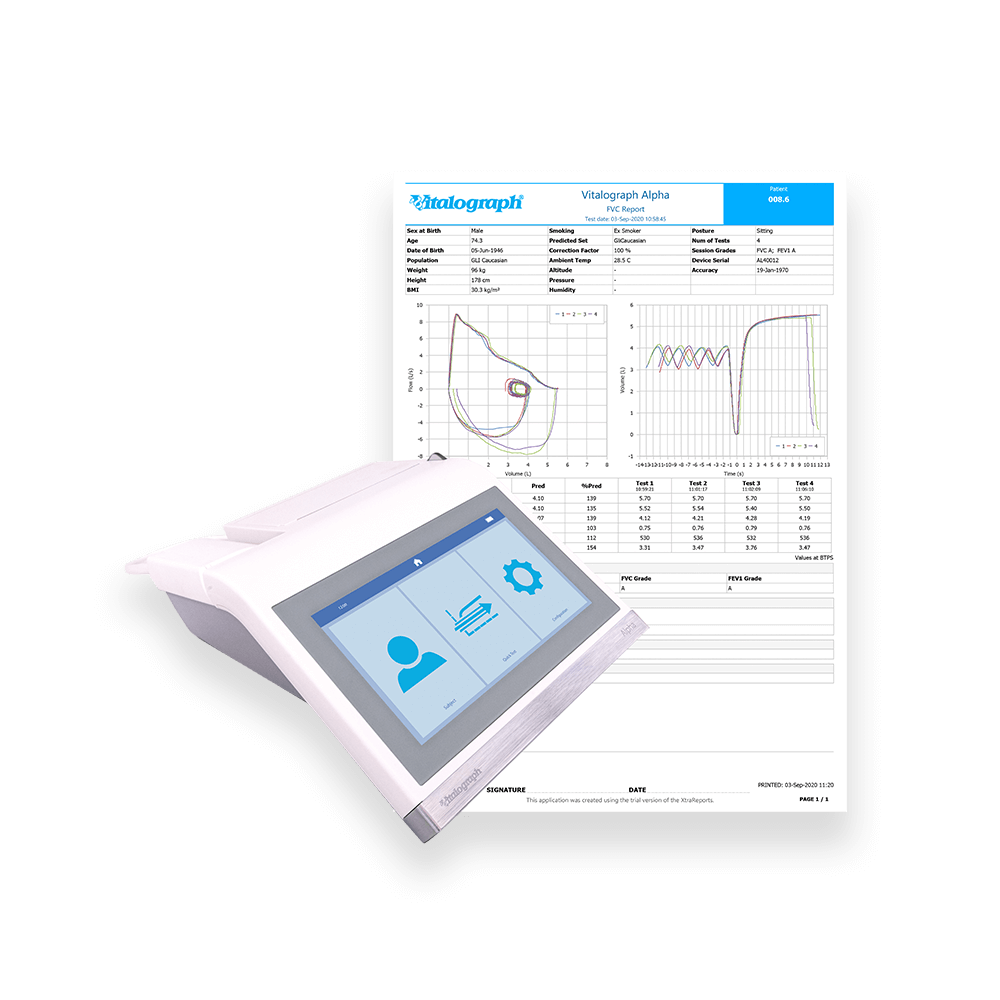
The Alpha encompasses compliance with the ATS/ERS 2019 Technical Standards, multiple testing options, quick loading integrated thermal printer and direct links to Vitalograph Device Studio to generate PDF reports. Integration with Spirotrac 6® respiratory diagnostic software enables test data to be centralised for detailed reporting and trending. Vitalograph Connect provides versatile connectivity to your electronic medical records via HL-7 and GDT, taking spirometry testing to the next level. Features a 7” high-resolution touch-screen with an intuitive user-friendly interface.
The Alpha Spirometer comes with:
- Vitalograph Device Studio
- Vitalograph Connect
- USB cable
- Ethernet cable
- Medically approved power supply with country-specific plug
- Carry case
This device comes with a Free 5-Year Warranty including unlimited technical support when registered within 30-days of purchase.
Vitalograph provides free certified training for all of its respiratory diagnostic solutions. Register here.
Click here for reimbursement codes for Vitalograph Devices in the USA.
Be free to help your patients perform Spirometry
The Alpha has an integrated V-Core™ flowhead, allowing you to focus on helping your patients achieve their best spirometry test, confident that you are capturing accurate and reproducible results.
Find out more about V-Core flowhead technology
Product code: 69002
- Meets 2019 ATS/ERS Technical Standards for accuracy and test quality.
- Supports full feature Spirometry Testing with over 60 available parameters.
- Utilising proven, highly accurate, robust, and V-Core™ measuring technology.
- Fast integrated thermal printer – No PC required.
- Lowest running costs & environmentally friendly: no need for costly disposable sensors, turbines, spirettes or flow tubes.
- Save time & money on cleaning using Vitalograph BVFs with validated cross-contamination efficiency >99.999%, protecting the device, patient, and operator.
- Instant quality feedback using the latest test/session acceptability, usability and repeatability criteria.
- Automatic interpretation based on ATS/ERS & GOLD standards.
- Ensure accurate results with quick and easy calibration verification routines as per 2019 ATS/ERS Technical Standards.
- Fast evaluation of results using latest GLI spirometry predicted equations with LLN, %Pred and Z-scores. Other predicted equations are selectable.
- Direct connectivity with Vitalograph Device Studio to generate PDF reports and receive the latest firmware updates.
- Integration with Spirotrac 6® respiratory diagnostic software enables test data to be centralised for detailed reporting and trending.
- Share test data easily with compatible Electronic Health Records (EHR) and Electronic Medical Records (EMR) systems through HL7, GDT.
| Vitalograph Model 6000 Alpha | |
|---|---|
| Flow detection principle | Fleisch type pneumotachograph |
| Essential performance test limits | Flow Accuracy ±10% or ±20 L/min, whichever is greater |
| Back pressure | Less than 0.1kPa/L/second @ 14L/s, complies with ATS/ERS 2019 |
| Volume detection | Flow integration sampling @ 100Hz |
| Maximum displayed volume | 10 L |
| Volume accuracy | +/- 2.5% ATS/ERS 2019, ISO 26782:2009 |
| Voltage/Frequency | Power Supply: Input 100-240V, 50/60HZ, Output 12V, 1.5A, Battery: 7.2V, 2.2Ahr |
| Accuracy when operated in temperature range conditions | Flow ±10%, Max. flow rate ±16 L/s, Min. flow rate ±0.02 L/s |
| Operating temperature range | ISO 26782 limits: 17–37ºC Design limits: 10–40ºC |
| Operating humidity range | 30%–75% |
| Ambient pressure range | 850hPa–1060hPa |
| Performance standards met by Vitalograph ALPHA | ATS/ERS 2019, ISO 23747:2015 & ISO 26782:2009 |
| Safety standards | EN 60601-1:2006 +A1:2013 +A2:2021 |
| QA/GMP standards | EN ISO 13485, FDA 21 CFR 820, SOR/98-282 & Japan’s PMD Act |
| Dimensions | 204 mm (length) x 253 mm (width) |
| Weight | 1.5 kg net |
| Communications | USB (Device Studio & Spirotrac 6), Ethernet/WiFi (Connect) |
| EMR Connectivity | HL7, GDT via Vitalograph Connect |
| Device Classification | IIa per Annex IX of the MDD |
| Test parameters measured | VC Test: VC, EVC, IVC, TV, IRV, ERV, IC FVC Test: VC, FVC, FIVC, FIVC/FVC, FEV0.5, FEV0.5/FVC, FEV0.75, FEV0.75/FVC, FEV1, FEV1R, FEV1/VC, FEV1/EVC, FEV1/IVC, FEV1/FVC, FEV1/FIVC, FEV1/FEV6, FEV1/PEF, FEV3, FEV3/VC, FEV3/FVC, FEV6, PEF L/s, PEF L/min, FEF25, FEF50, FEF75, FEF0.2-1.2, FEF25-75, FEF25-75/FVC, FEF75-85, FIV1, FIV1/FVC, FIV1/FIVC, PIF L/s, PIF L/min, FIF25, FIF50,FIF75, FIF50-FEF50, FEF50-FIF50, MVVind, FMFT, FET, FRC, TV, RV, TLC, IRV, ERV, IC, Rind, ELA, tPEF, Text, EV, EV/FVC, |
- Vitalograph Alpha Spirometer: Instructions for Use (Issue 6)
- Vitalograph Alpha Spirometer: Instructions for Use (Issue 5)
- Vitalograph Alpha Spirometer: Instructions for Use (Issue 4)
- Vitalograph Device Studio Software: Instructions for Use
- Vitalograph Alpha Spirometer: Brochure
- Vitalograph Alpha Spirometer: Instructions for Use (Issue 3)
Product Specification
| ALPHA | ALPHA Touch | ALPHA Touch with Spirotrac | |
|---|---|---|---|
| Tests: | |||
| VC | |||
| FVC | |||
| Post BD | |||
| Total Parameters Available | 9 | 56 | 60 |
| Quality Measures & Incentives: | |||
| Predicted Values | |||
| Interpretation | |||
| Quality Prompts | |||
| Incentives | |||
| Data Exchange & Printing: | |||
| In-Built Printer | |||
| PDF Reports (V=Vitalograph Reports, S=Spirotrac) | V | V | S/V |
| Bi-directional Data Exchange with EMRs | |||
| General Features: | |||
| Touch Screen | |||
| Color Display | |||
| Internal Battery | |||
| Full Database | |||
FAQs
Where a new BVF is used for each subject, only the flowhead surface which comes into contact with subjects being tested should require low level cleaning. For cleaning instructions refer to our Alpha Cleaning Instructions.
You have the ability to perform a simple calibration verification routine prior to undertaking testing, in accordance with international spirometry standards and guidelines (ATS/ERS).
VC Test: VC, EVC, IVC, TV, IRV, ERV, IC
FVC Test: VC, FVC, FIVC, FIVC/FVC, FEV0.5, FEV0.5/FVC, FEV0.75, FEV0.75/FVC, FEV1, FEV1R, FEV1/VC, FEV1/EVC, FEV1/IVC, FEV1/FVC, FEV1/FIVC, FEV1/FEV6, FEV1/PEF, FEV3, FEV3/VC, FEV3/FVC, FEV6, PEF L/s, PEF L/min, FEF25, FEF50, FEF75, FEF0.2-1.2, FEF25-75, FEF25-75/FVC, FEF75-85, FIV1, FIV1/FVC, FIV1/FIVC, PIF L/s, PIF L/min, FIF25, FIF50, FIF75, FIF50-FEF50, FEF50-FIF50, MVVind, FMFT, FET, FRC, TV, RV, TLC, IRV, ERV, IC, Rind, ELA, tPEF, Text, EV, EV/FVC.
Running costs are low for the Alpha as no expensive disposable sensors, adaptors or flow tubes are required. The simple, validated hygiene system using Bacterial Viral Filters means that keeping the device clean is easy and inexpensive.
The Alpha comes with 2 spare rolls of thermal printer paper. Replacements are available in packs of 10 rolls. This ensures you never run short, at a cost effective price point, when ordered directly from Vitalograph.
Talk to Us
For information on any of our products, click the inquiry button, enter your query and we will get back to you.
Talk to Us
For information on any of our products, click the inquiry button, enter your query and we will get back to you.